
- 997556699
- [email protected]
medical devices
21
Oct
Oct

Insight on Perú IVD and medical device regulations
by Pharma Consulting | medical devices, registration | 0 Comment
In Perú, the General Directorate of Medicines, Supplies, and Drugs (DIGEMID) oversees the IVD and medical device market. This agency registers devices, issues operation licenses, inspects manufacturing facilities and warehouses, monitors product testing, and drafts policies and laws regarding medical devices imported to and sold in Perú. Local regulations and medical device classification DIGEMID classifies
13
Jul
Jul
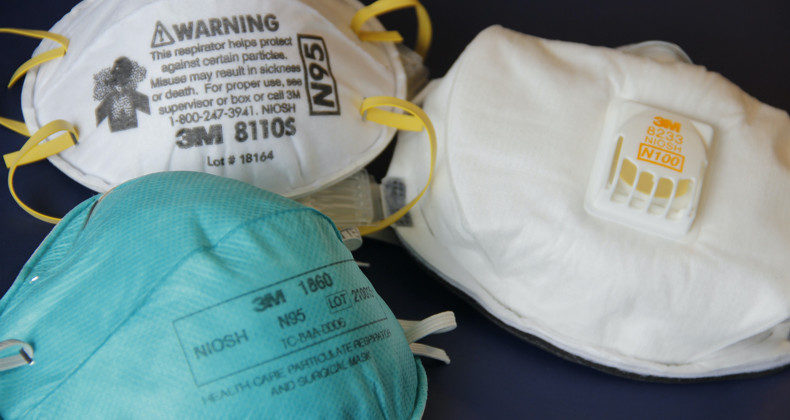
N95, FFP2 and KN95 medical respirators must be registered with DIGEMID
by Pharma Consulting | COVID-19, medical devices | 0 Comment
The Directorate of Medicines, Supplies, and Drugs - DIGEMID, has established the mandatory registration of respirators for medical use as of January 1, 2021. This mandate affects institutions and pharmaceutical establishments that import, store, manufacture, or market N95, KN95, FFP2, FFP3 respirators for medical use, and the like, as they are considered class I medical
19
Oct
Oct

DIGEMID reduced the evaluation period for the registration of medical devices in Peru
by Pharma Consulting | Digemid, medical devices | 0 Comment
The National Authority of Pharmaceutical Products, Medical Devices, and Health Products has reduced the maximum period of evaluation for the inscription and reinscription of medical devices in the Peruvian sanitary registry. Medical Devices (MD) Class I, low risk: Up to thirty (30) calendar days. Medical Devices (MD) Class II, moderate risk: Up to sixty (60)
20
Sep
Sep

DIGEMID allows direct selling of medical devices to patients, healthcare institutions, professionals, and retailers
According to D.R. No. 033-2017-DIGEMID-DG-MINSA, published early this year, DIGEMID issued a list of medical devices that may be marketed directly to end-users. This document does not list every medical device but establishes a criterion based on MD risk classification. Medical Devices Class I, II, III, and IV Laboratories and droguerías (pharmaceutical establishments) may directly
20
Aug
Aug
List of medical devices that can be marketed directly to end-users in Perú
by Pharma Consulting | Digemid, medical devices | 0 Comment
The Directorate of Medicines, Supplies, and Drugs - DIGEMID, issued a list of medical devices that can be marketed directly to end-users. In order to acquire a particular product, users must present a medical prescription, if required. Biomedical Equipment Class I (low risk), class II (moderate risk), class III (high risk), and class IV (critical