
- 997556699
- [email protected]
registration
26
Apr
Apr

Regulatory aspects of dietary products and sweeteners in Peru
by Pharma Consulting | dietary supplements, registration | 0 Comment
Regulatory aspects of dietary products and sweeteners The General Directorate of Medicines, Supplies, and Drugs (DIGEMID), during the first "Virtual technical meeting on the registration process of nutritional supplements and sweeteners", established some criteria to be taken into account by users interested in registering these products. Requirements for the health registration of dietary supplements (nutritional)
04
Jan
Jan
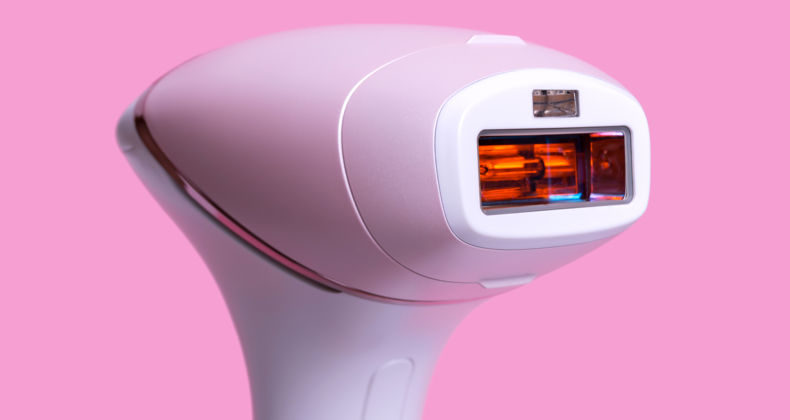
Registration of laser, IPL and radiofrequency equipment for use in aesthetics in Peru
by Pharma Consulting | dispositivos médicos, medical devices, registration, registro sanitario | 0 Comment
Laser, IPL and radiofrequency equipment for use in aesthetics A device for use in aesthetic medicine is classified biomedical, active, non-invasive, or minimally invasive device that acts through the administration or exchange of energy in the form of non-ionizing radiation, to or from the human body. Most of the equipment in this category belongs to
25
Dec
Dec

Multivitamins slow cognitive aging in population over age 65
by Pharma Consulting | pharmaceutical, registration | 0 Comment
Preliminary results of a randomized study with adult patients older than 65 years. The researchers found that over a 3-year period of treatment, patients who received multivitamins such as vitamin B12, folate, vitamin D, and other micronutrients reduced fared better than those who received placebo. Investigators estimated that multivitamins were slowing cognitive aging by 60%
21
Oct
Oct

Insight on Perú IVD and medical device regulations
by Pharma Consulting | medical devices, registration | 0 Comment
In Perú, the General Directorate of Medicines, Supplies, and Drugs (DIGEMID) oversees the IVD and medical device market. This agency registers devices, issues operation licenses, inspects manufacturing facilities and warehouses, monitors product testing, and drafts policies and laws regarding medical devices imported to and sold in Perú. Local regulations and medical device classification DIGEMID classifies