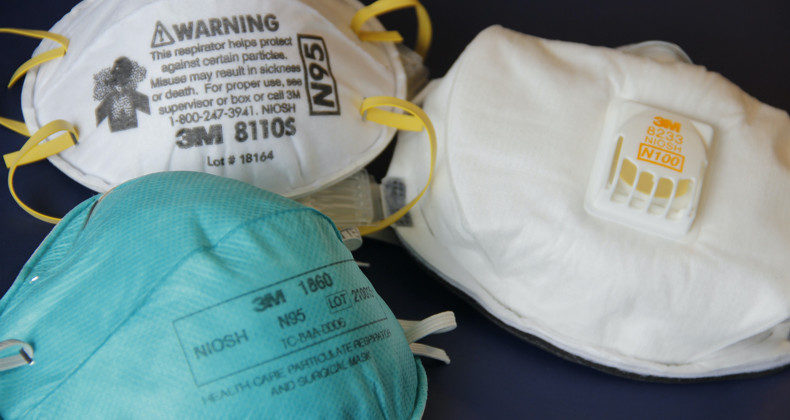
The Directorate of Medicines, Supplies, and Drugs – DIGEMID, has established the mandatory registration of respirators for medical use as of January 1, 2021. This mandate affects institutions and pharmaceutical establishments that import, store, manufacture, or market N95, KN95, FFP2, FFP3 respirators for medical use, and the like, as they are considered class I medical devices.
Individuals and businesses not registered with DIGEMID will not be permitted to import and distribute this kind of products.
Deadlines
This new requirement will take effect on January 1, 2021.
Requirements
The requirements to register respirators for medical use are established by DIGEMID. You can learn more about the procedure by clicking here.
Respirator models exempt from registration
N95 Non-medical respirators
The N95 respirator is a respiratory protection device designed to achieve a good facial fit and very efficient filtration of airborne particles. The N95 designation means that, when carefully tested, the respirator blocks at least 95 percent of very small particles (0.3 microns). If properly fitted, the filtration capabilities of N95 respirators exceed those of surgical masks.

KN95 Non-medical respirators
The KN95 respirator is not resistant to oily airborne particles. It is recommended to use it in conjunction with a face shield.
FFP2, FFP3 Non-medical particulate respirators
FFP2 and FFP3 particulate respirators are not resistant to fluids. It is recommended to use them in conjunction with a face shield.
Three-ply mask
Also called a three-fold mask or disposable mask, it is a medical material that covers the mouth and nose, and provides a barrier to minimize the direct transmission of infectious agents between medical personnel and patients.

Contact us at 997556699 or [email protected] to learn more about medical respirators registration.
Source: DIGEMID