
- 997556699
- [email protected]
registration
28
Apr
Apr

Characteristics of the regulation of medical devices and equipment in Peru
by Pharma Consulting | biomedical equipment, medical devices | 0 Comment
Regulatory Authority In Peru, the General Directorate of Medicines, Supplies, and Drugs (DIGEMID) is the National Authority that oversees the medical devices, equipment, pharmaceutical products, personal care, and health products market. Summary of Peruvian regulatory legislation Is it necessary to obtain authorization to operate as a company prior to marketing restricted products? Yes, according to
04
Jan
Jan
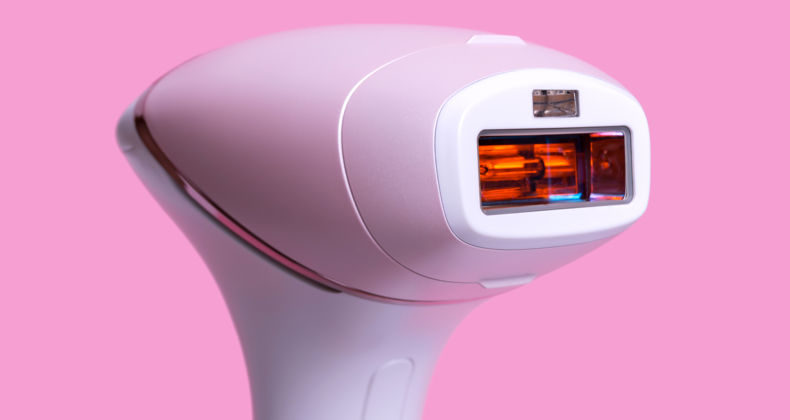
Registration of laser, IPL and radiofrequency equipment for use in aesthetics in Peru
by Pharma Consulting | dispositivos médicos, medical devices, registration, registro sanitario | 0 Comment
Laser, IPL and radiofrequency equipment for use in aesthetics A device for use in aesthetic medicine is classified biomedical, active, non-invasive, or minimally invasive device that acts through the administration or exchange of energy in the form of non-ionizing radiation, to or from the human body. Most of the equipment in this category belongs to
19
Oct
Oct

DIGEMID reduced the evaluation period for the registration of medical devices in Peru
by Pharma Consulting | Digemid, medical devices | 0 Comment
The National Authority of Pharmaceutical Products, Medical Devices, and Health Products has reduced the maximum period of evaluation for the inscription and reinscription of medical devices in the Peruvian sanitary registry. Medical Devices (MD) Class I, low risk: Up to thirty (30) calendar days. Medical Devices (MD) Class II, moderate risk: Up to sixty (60)